For Immediate Release —- (Washington D.C. April 23, 2026) On April 20, 2026 Community Voices for 340B (CV340B) submitted comments to the Health Resources and Services Administration (HRSA) raising serious concerns that the proposed 340B rebate model could disrupt care delivery for millions of patients. CV340B’s submission was part of a significant wave of stakeholder engagement, with an estimated 5,500 comments submitted alongside it, underscoring the widespread concern across the 340B community. In its comments, CV340B highlights risks to provider cash flow, increased administrative burden, and a lack of manufacturer transparency, while offering actionable solutions to ensure the program continues to meet its mission of supporting access to care for vulnerable populations.
To download: CV340B Comments on 340B Rebate RFI
Text to follow
******
April 20, 2026
SUBMITTED ELECTRONICALLY VIA HTTPS://WWW.REGULATIONS.GOV
Chantelle Britton
Director
Office of Pharmacy Affairs
Office of Special Health Initiatives
Health Resources and Services Administration
5600 Fishers Lane, Mail Stop 10W29
Rockville, MD 20857
RE: HHS Docket No. HRSA–2026–03042: Community Voices for 340B Comments in Response to Request for Information: 340B Rebate Model Pilot Program
Introduction
Community Voices for 340B (CV340B) thanks the Health Resources and Services Administration (HRSA) for the opportunity to respond to HRSA’s Request for Information (RFI) examining the potential impacts of a 340B Drug Pricing Rebate Program. It is paramount that HRSA preserves the intent and integrity of the 340B drug pricing program (340B program) as it considers changes to the way the program has operated for the last 30 years.
CV340B Who We Are
Community Voices for 340B (CV340B) is a grassroots organization that seeks to raise awareness of the important role that the 340B program plays in protecting and improving health care access and the quality of care for communities nationwide. Over the last ten years, CV340B has been the only national organization that provides a space for all 340B stakeholders to come together to #Defend340B. CV340B’s five Regional Advocacy Groups has over six hundred engaged individual advocates dedicated to defending the 340B program.
Why We Care
Working closely with its “covered entity advocates”, which includes representatives from Hospitals, Federally Qualified Community Health Centers, Ryan White Clinics, Pharmacies and other 340B stakeholders, CV340B learns firsthand about the immense value of the 340B program to the patients served by covered entities. Advocates partner with CV340B to educate their patients, staff, community and policymakers about the importance of the 340B Program. Intended to stretch scarce federal resources to reach more patients and provide more services, the 340B program provides financially strapped covered entities with a stable source of savings and revenue that they use to underwrite the cost of daily operations and patient care. But that stable funding source depends on covered entities receiving their 340B discounts at the point of sale. A shift away from upfront discounts puts this fragile financing system at risk, ultimately harming patients who depend on covered entities for care that they cannot afford to get elsewhere. When covered entity advocates were asked by CV340B in a recent survey about the perceived advantages of moving to a rebate-based model, 38 respondents reported they saw no advantage. Many respondents simply said, ‘There is none.’ One of those respondents elaborated on the sentiment of the group stating:
There are no advantages for a [covered entity] moving to a rebate-based model. The only advantage is to pharma [the pharmaceutical industry]. CEs will have to front mon(ies) to pharma in the form of the retail price of a medication–and pharma will have discretion whether to approve or deny the rebate to the CE. This is not sustainable.
None of the respondents expressed support for the rebate model.
What Is Included in the RFI
CV340B appreciates HRSA’s thoroughness in gathering information on the 340B rebate model from interested stakeholders. As explained in more detail below, CV340B has serious concerns with the proposed model, especially with respect to the financial and operational impact on covered entities, patient harm, and the lack of manufacturer transparency and accountability.
Cost to Covered Entities
Covered entities will incur considerable cost as a result of a 340B rebate program. The anticipated impact on operating margins, drug costs, wholesaler payments, cash flow, and patient care are just a few of covered entities’ most pressing concerns with the proposed transition away from an upfront discount.
Operational Impact
The increased operational burden of managing a 340B rebate will cripple small covered entities. Safety-net hospitals, community health centers, Ryan White clinics, and other 340B covered entities operate on razor thin margins. An analysis of federally regulated Uniform Data Set indicates that Federally Qualified Community Health Centers (FQHCs) operate on a daunting -2% operating margin.[1] Hospitals across the country continue to struggle with chronic Medicaid and Medicare underpayments, high levels of inflation, rising costs of health insurance premiums, and rapid growth in drug costs.[2] Receiving 340B discounts upfront allows covered entities to mitigate the impact of these environmental factors and to invest their savings immediately in patient care. HRSA’s proposed rebate model would significantly destabilize the current financing system for safety-net providers in the U.S. The shift to a rebate model would disrupt cash flow stability, increase dependance on credit lines, and force staffing and service decisions that will directly impact patients.
Rising Drug Costs
The unsustainable rise in drug costs – and the challenges to patient care created by rising drug costs – will be exacerbated by a 340B rebate model. The median cost for a new drug coming to market in 2024 was $370,000, more than double the cost in 2021.[3] Beqvez, a drug that treats the bleeding disorder hemophilia and manufactured by Pfizer, hit the market in 2024 with a whopping list price of $3,500,000 per dose, making it completely unaffordable for families trying to protect their loved ones from a fatal bleed.[4] The astounding increase in drug prices does not end with new drugs entering the market. The Office of the Assistant Secretary for Planning and Evaluation found that, in 2022, more than 4,200 drugs had an average price increase of 15.2%, nearly double the rate of inflation in the same year.[5] The 340B program was designed to combat these rising and often exorbitant drug costs. By shifting to a rebate model, covered entities would be forced to bear the full brunt of these high drug costs before receiving any relief from manufacturers. Covered entities are rightfully concerned that if drug companies are willing to price their products far beyond what is considered a reasonable or fair price, they may be just as willing to withhold or delay payment of 340B rebates. The rebate model, therefore, introduces both increased financial risk and administrative burden to the covered entities attempting to make these drugs affordable for their patients.
Wholesaler Payments and Cash Flow Impacts
The terms and conditions of covered entities’ current wholesaler agreements were all negotiated based on the assumption of an upfront discount. While specific contract terms are confidential, our covered entity advocates report that wholesaler payment timelines vary from a 30-day payment period to a same-day or prepay arrangement with an average 17-day payment timeline. Wholesaler payment terms are determined in part by cash on hand and previous credit history. While some covered entities have a standard three-month cash on hand, others are operating on less than a day. For those covered entities with prepay or same day wholesaler repayment terms and less than one day cash on hand, a rebate model could force them out of business. Wholesaler payment timelines are structured around predictable pricing. The rebate model creates uncertainty by requiring covered entities to pay full price for the drugs they order and giving them little control over when the rebates for those drugs arrive. They can only hope to receive the rebates from manufacturers in a timely manner. Even the most lenient wholesaler timelines become untenable when manufacturers unilaterally decide to deny rebates.
Patient Harm
Due to the inevitable financial instability caused by a potential 340B rebate model, patient access to affordable medication and services will be greatly reduced. Our covered entity advocates tell us that they are already bracing for the changes they will have to make to compensate for the delay in receiving a rebate. Some of these anticipated changes include limiting access to high-cost medications for those patients who might have another drug option; reducing services for which 340B was originally intended to support, such as transportation, medication adherence support, and case management; and scaling back community-facing programs like heart health programs and diabetes support services. In a recent survey of our advocates, 36.1% of respondents reported that a rebate program would result in the immediate closure of a facility or clinic. One respondent said, “Our cancer center would close immediately.”
What’s Missing: Manufacturer Audits and Transparency
Manufacturers assert that the rebate model will reduce duplicate discount risks and increase transparency within the 340B program. Noticeably missing from this rationale is an acknowledgement that manufacturer overcharges of covered entities is not being adequately addressed.
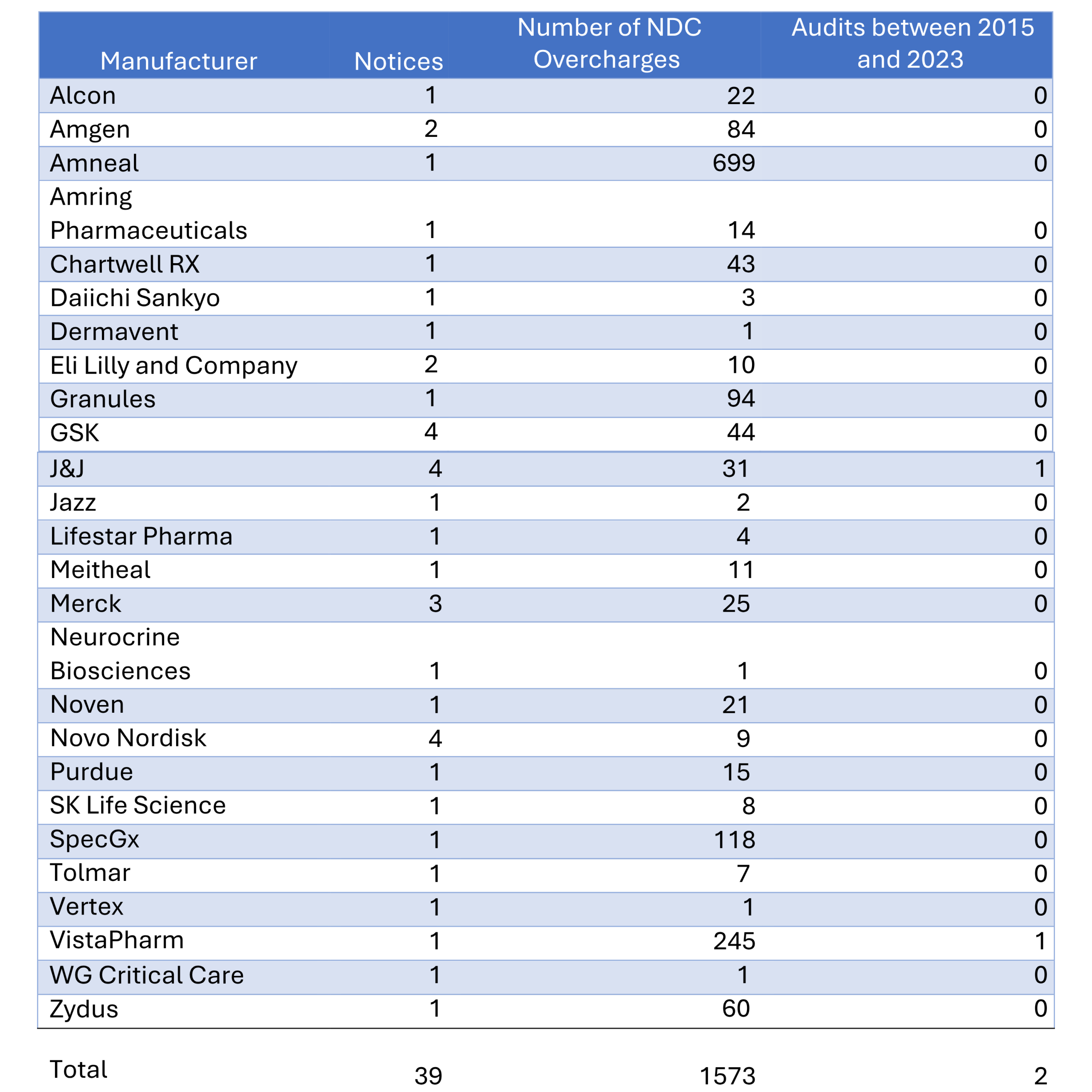
HRSA should establish an automatic review of a manufacturer when there is reasonable evidence that it overcharged covered entities. The purpose of the review should be to determine whether the manufacturer should be audited. Enforcement mechanisms must be timely, transparent, and meaningful to ensure compliance. Covered entities are being audited at nearly forty times the rate of manufacturers. In 2023, 26 manufacturers issued 39 individual notices of overcharge to covered entities on 1,573 distinct NDCs.[6] Yet, only two drug manufacturers out of the 26 manufacturers who reported overcharging covered entities have been audited by HRSA.3 HRSA’s audit program is not solving the overcharge problem. In 2023, Genentech issued a notice to covered entities detailing a TEN-YEAR period from 2011 to 2021 in which it overcharged covered entities for the orphan drug Klonopin (clonazepam). Like many other drug companies, Genentech has not been audited by HRSA in the past 10 years. Another problem covered entities face is securing repayments from manufacturers to rectify overcharges. The administrative burden of tracking and seeking refunds from manufacturers for overcharges rests entirely on covered entities. Drug companies have access to purchasing records and could easily issue refunds to covered entities automatically. However, in what can only be described as a cost-saving measure, drug companies require covered entities to submit a refund request or credit claim for each overcharged NDC.
| Manufacturer | Notices | Number of NDC Overcharges | Audits between 2015 and 2023 |
| Alcon | 1 | 22 | 0 |
| Amgen | 2 | 84 | 0 |
| Amneal | 1 | 699 | 0 |
| Amring Pharmaceuticals | 1 | 14 | 0 |
| Chartwell RX | 1 | 43 | 0 |
| Daiichi Sankyo | 1 | 3 | 0 |
| Dermavent | 1 | 1 | 0 |
| Eli Lilly and Company | 2 | 10 | 0 |
| Granules | 1 | 94 | 0 |
| GSK | 4 | 44 | 0 |
| J&J | 4 | 31 | 1 |
| Jazz | 1 | 2 | 0 |
| Lifestar Pharma | 1 | 4 | 0 |
| Meitheal | 1 | 11 | 0 |
| Merck | 3 | 25 | 0 |
| Neurocrine Biosciences | 1 | 1 | 0 |
| Noven | 1 | 21 | 0 |
| Novo Nordisk | 4 | 9 | 0 |
| Purdue | 1 | 15 | 0 |
| SK Life Science | 1 | 8 | 0 |
| SpecGx | 1 | 118 | 0 |
| Tolmar | 1 | 7 | 0 |
| Vertex | 1 | 1 | 0 |
| VistaPharm | 1 | 245 | 1 |
| WG Critical Care | 1 | 1 | 0 |
| Zydus | 1 | 60 | 0 |
| Total | 39 | 1573 | 2 |
https://www.hrsa.gov/opa/manufacturer-notices
It is difficult to understand how the rebate model would improve 340B program integrity when the model does nothing to advance manufacturer transparency and accountability.
To address this glaring omission, CV340B recommends that HRSA require manufacturers to regularly submit standardized, publicly available data before implementing a 340B rebate model. Such data should include total value of all historical overcharges committed by the manufacturer, drug price increases that exceed the rate of inflation for each NDC sold by the manufacturer, disclosure of all manufacturer-supported patient advocacy groups, and disclosure of all manufacturer-supported studies and articles relating to the 340B program.
There are good reasons to expand the rebate initiative to include these manufacturer data submission requirements. Manufacturers impose tremendous administrative burden on covered entities as previously mentioned. It is only fair that manufacturers be required to provide the total number and value of reported overcharges to HRSA.
Penny pricing in the 340B program occurs when a manufacturer raises the price of drugs far beyond the rate of inflation. The purpose of this penalty is to protect covered entities from such dramatic price increases. If manufacturers did not habitually raise their prices faster than the rate of inflation, the 340B discount on brand drugs would be a standard 23%. HRSA, covered entities and the general public are entitled to information relating to these price increases, including the manufacturers’ justification for the increases.
During a recent review of the submitted RFI comments on the rebate model, the covered entity community noticed that Patients Rising, a manufacturer-funded patient advocacy group, flooded HRSA with AI-generated letters supporting the model.[7] More than half of the initial 300 RFI comments were identical letters submitted in alphabetical order starting with commenters whose names started with the letter ‘a,’ but were not otherwise identifiable. Whether these letters came from actual patients is an open question. This “astro-turf” campaign is strong evidence of the egregious quid-pro-quo overreach of manufacturer’s financial support of patient advocacy groups. CV340B has spent weeks and countless hours educating and preparing covered entities and their patients on the Request for Information from HRSA on the 340B rebate model. For too long, drug manufacturers have hidden behind organizations that have purportedly represented “patients” while doing the bidding of drug companies without their members knowledge.
Furthermore, drug manufacturers’ ability to buy research to spread misinformation about the 340B program continues to threaten the care that patients receive from covered entities. Funding studies through consulting firms and think tanks which use selective framing of research questions and incomplete datasets leads to research that spreads misinformation that influences policy makers who rely on the seemingly credible data. Lack of transparency of funding sources and methodologies increases covered entity administrative burden, and financial uncertainty due to increased policy threats and restrictions and causes reputational harm to covered entities who attempt to defend the 340B program from these false narratives. Reinforcing 340B misinformation through manufacturer funded research is not only a policy concern when considering a rebate model, it has real tangible consequences for patients’ access and health outcomes.
It is CV340B’s belief that increased drug manufacturer transparency should start with a disclosure of all manufacturer engagements with patient advocacy groups, researchers, and academic scientists.
Independent Clearinghouse for Duplicate Discounts
There are other reasons to be skeptical of manufacturers’ claim that the rebate model is needed to reduce duplicate discount risks. First, the risks are overstated. HRSA has essentially eliminated the Medicaid fee-for-service duplicate discount risk through its Medicaid Exclusion File solution. Implementation of the Inflation Reduction Act’s Medicare price reduction provisions is still in its infancy. It is too early to declare the existence of a Medicare/340B duplicate discount problem.
Second, there are other options for managing potential duplicate discounts without inflicting harm to the safety net. CV340B supports the creation of a neutral clearinghouse. An independent clearinghouse could prevent duplication and reduce the administrative and financial burdens placed on covered entities compared to a rebate model. Any clearinghouse should only be used to prevent federally prohibited 340B duplicate discounts involving Medicaid rebates and Medicare maximum fair price and inflationary rebates. It should not be used to address 340B duplicate discounts involving commercial claims.
Conclusion
The 340B program is currently operating as intended. The program’s effectiveness in generating a stable funding source for covered entities would be in grave jeopardy if a 340B rebate model is established. Shifting the financial risk from multinational, for-profit conglomerates to the non-profit safety net makes no sense and would undermine access to affordable healthcare for patients. CV340B urges HRSA not to capitulate to drug company pressure to launch a 340B rebate model. HRSA should instead stand with patients and the American people. We welcome continued dialogue with HRSA and are ready to support policies that protect the 340B program and the patients it serves.
For more information, please contact Rhiannon Marshall Klein, Executive Director at Rhiannon.Marshall@cv340B.org or 616.485.9378.
Sincerely,
Rhiannon Klein
Executive Director
Community Voices for 340B
[1] https://data.hrsa.gov/topics/healthcenters/uds/overview
[3] https://trial.medpath.com/news/05f77dd2c80303fb/u-s-drug-prices-double-as-pharmaceutical-industry-shifts-focus-to-rare-diseases.
[4] https://www.reuters.com/business/healthcare-pharmaceuticals/prices-new-us-drugs-doubled-4-years-focus-rare-disease-grows-2025-05-22/#:~:text=The%20percentage%20of%20drugs%20launched,price%20of%20$22%2C500%20a%20year.
[5] https://aspe.hhs.gov/reports/changes-list-prices-prescription-drugs.
[6] https://www.hrsa.gov/opa/manufacturer-notices.
[7] https://340breport.com/patients-rising-ends-outreach-campaign-after-340b-report-highlights-identical-hrsa-340b-rfi-submissions/.